 Dedicated to all moms in the world
Dedicated to all moms in the world
Rule 2, omit O, gives C5H10
5 – 10/2 + 1 = 1 degree of unsaturation.
Look for 1 pi bond or aliphatic ring.
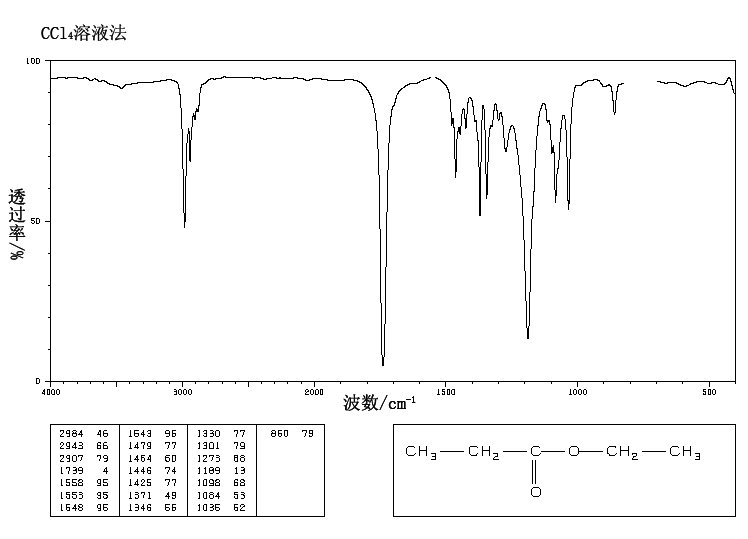
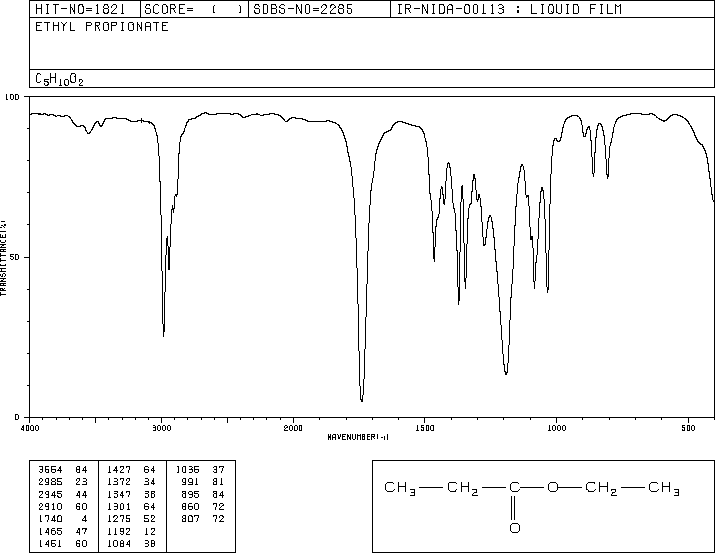


 This is the structure. See if you can assign the peaks on your…
This is the structure. See if you can assign the peaks on your…View original post 107 more words
 Dedicated to all moms in the world
Dedicated to all moms in the world




 This is the structure. See if you can assign the peaks on your…
This is the structure. See if you can assign the peaks on your…View original post 107 more words

Active pharmaceutical ingredients (APIs) are frequently delivered to the patient in the solid state as part of such dosage forms as tablets, capsules, etc.In this context the ability to deliver the drug to the patient in a safe, efficacious and cost-effective way depends largely on the physicochemical properties of the APIs in the solid state, and ……..read more
| The ultimate goal of drug synthesis is to scale up from producing milligram quantities in a laboratory to producing kilogram to ton quantities in a plant, all while maintaining
all at |
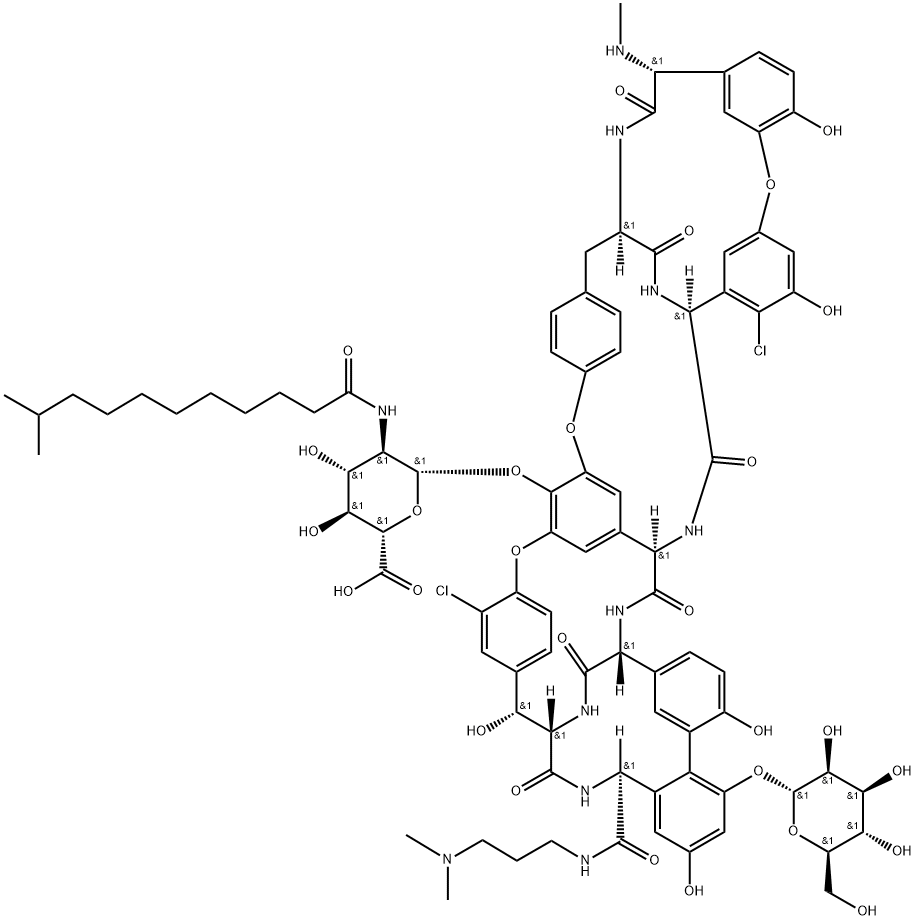
| CAS No. | 171500-79-1 |
| Chemical Name: | Dalbavancin |
| Synonyms: | MDL 63397;Dalbavancin |
| CBNumber: | CB41028737 |
| Molecular Formula: | C88H100Cl2N10O28 |
| Formula Weight: | 1816.71 |
CHICAGO, Nov 26, 2013 (GLOBE NEWSWIRE via COMTEX) — Durata Therapeutics, Inc. DRTX +6.48% today announced that the New Drug Application (NDA) for its investigational drug, Dalvance (dalbavancin hydrochloride) for injection, has been accepted for priority review by the U.S. Food and Drug Administration (FDA) with an action date of May 26, 2014. Durata is seeking FDA approval of Dalvance(TM) for the treatment of patients with acute bacterial skin and skin structure infections (ABSSSI) caused by susceptible Gram-positive microorganisms, including MRSA (methicillin resistant Staphylococcus aureushttp://www.drugs.com/nda/dalbavancin_131126.html

View original post 663 more words

VBL Therapeutics announced today that the U.S. Food and Drug Administration (FDA) has granted Fast Track designation to its lead oncology drug VB-111, for prolongation of survival in patients with recurrent glioblastoma multiforme (rGBM).
VB-111 is the first highly targeted anti-angiogenic agent for the specific inhibition of tumor vascular growth to use VTS™™, our proprietary platform technology, for cancer therapy. VB-111 is an IV-administered anti angiogenic agent that works in a manner akin to a “biological knife” to destroy tumor vasculature, thus cutting off blood vessels feeding the tumor.
VB-111 has shown…
View original post 158 more words
Teva Announces Additional Regulatory Exclusivity for TREANDA® (Bendamustine HCI) for Injection
Orphan Designation combined with pediatric extension provides regulatory exclusivity through April 2016 for indolent B-cell non-Hodgkin lymphoma indication
JERUSALEM, November 27, 2013 –(BUSINESS WIRE)–Teva Pharmaceutical Industries Ltd. (NYSE: TEVA) today announced that the U.S. Food and Drug Administration (FDA) has granted orphan drug exclusivity for TREANDA through October 2015 for indolent B-cell non-Hodgkin lymphoma (iNHL) that has progressed during or within six months of treatment with rituximab or a rituximab-containing regimen.http://www.pharmalive.com/teva-announces-additional-regulatory-exclusivity-for-treanda
read my old post, contains synthesis
http://newdrugapprovals.wordpress.com/2013/09/19/fda-oks-tevas-injectable-treanda/

LEVETIRACETAM, etiracetam
(-)-(S)-α-ethyl-2-oxo-1-pyrrolidine acetamide
(−)-(S)-alpha-ethyl-2-oxo-1-pyrrolidineacetamide
CAS…102767-28-2
Crystals from ethyl acetate, mp 117°. [a]25D -90.0° (c = 1 in acetone). Soly (g/100 ml): water 104.0; chloroform 65.3; methanol 53.6; ethanol 16.5; acetonitrile 5.7. Practically insol in n-hexane. LD50 in male mice, male rats (mg/kg): 1081, 1038 i.v. (Gobert, 1990).
Mp: mp 117°C
View original post 7,573 more words